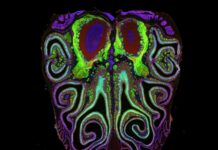
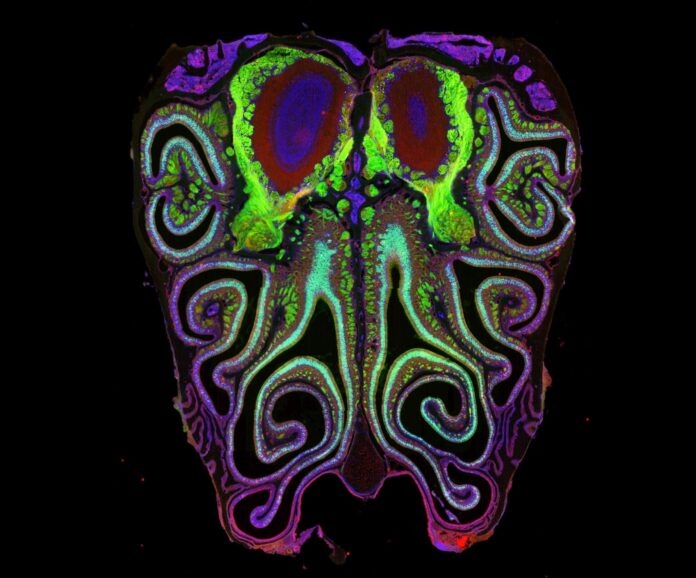
For decades, the science of smell remained a biological black box. While scientists had long mapped the precise arrangements of receptors for sight, hearing, and touch, the olfactory system was considered chaotic and random. That assumption has now been overturned.
Researchers at Harvard Medical School have unveiled the first detailed map of smell receptors in the nose, revealing a highly organized structure that challenges previous understanding. This discovery not only solves a 35-year-old mystery but also opens new pathways for treating the growing public health crisis of smell loss.
Breaking the Myth of Randomness
The study, led by Professor Sandeep (Robert) Datta of the Blavatnik Institute at Harvard Medical School, focused on mice, which share significant olfactory similarities with humans. The team analyzed approximately 5.5 million neurons from over 300 mice, combining single-cell sequencing with spatial transcriptomics to identify both the type of receptor each neuron expressed and its exact physical location.
The results were striking. Contrary to the long-held belief that smell receptors were distributed randomly, the neurons are arranged in tightly packed, horizontal stripes running from the top to the bottom of the nasal cavity. Each stripe corresponds to a specific type of smell receptor.
This organization is not isolated to the nose. The study found that this nasal map aligns perfectly with corresponding maps in the olfactory bulb of the brain. This structural symmetry suggests that scent signals travel from the nose to the brain in a highly ordered fashion, similar to how visual or auditory information is processed.
“Our results bring order to a system that was previously thought to lack order, which changes conceptually how we think this works,” said Datta, senior author of the study published in Cell.
Why It Took So Long to Find the Map
The complexity of the olfactory system made it difficult to map. While color vision relies on just three main receptor types, mice possess around 20 million olfactory neurons expressing more than 1,000 different receptor types. Each receptor is tuned to detect specific groups of odor molecules, creating a system of immense variety.
Early studies in the 1990s identified broad zones for receptors but lacked the resolution to see finer details, leading to the conclusion that distribution was largely random. It wasn’t until recent advances in genetic tools allowed for large-scale, high-resolution analysis that the hidden pattern could be revealed.
As Datta noted, “This is now arguably the most sequenced neural tissue ever, but we needed that scale of data in order to understand the system.”
The Chemical Blueprint: Retinoic Acid
The research also uncovered how this complex map is built during development. The team identified retinoic acid, a molecule that regulates gene activity, as the key architect.
A gradient of retinoic acid within the nose acts as a positional guide. Neurons determine which receptor to express based on their location relative to this chemical gradient. When researchers experimentally increased or decreased retinoic acid levels, the entire receptor map shifted up or down in the nose.
“We show that development can achieve this feat of organizing a thousand different smell receptors into an incredibly precise map that’s consistent across animals,” Datta explained.
These findings were corroborated by a separate study published in the same issue of Cell, led by Catherine Dulac, Xander University Professor at Harvard University, further validating the existence of this spatial code.
Implications for Health and Treatment
The discovery has profound implications for medicine. Loss of smell (anosmia) is increasingly common, affecting safety, nutrition, and mental health. Currently, there are limited treatments because the underlying biology was poorly understood.
Understanding this map is a prerequisite for developing effective therapies. Future treatments may include:
* Stem cell therapies designed to replace damaged neurons in their correct spatial locations.
* Brain-computer interfaces that bypass damaged nerves by stimulating the olfactory bulb directly.
* Targeted interventions for conditions linked to smell loss, such as depression and cognitive decline.
“Smell has a really profound and pervasive effect on human health, so restoring it is not just for pleasure and safety but also for psychological well-being,” Datta said. “Without understanding this map, we’re doomed to fail in developing new treatments.”
Conclusion
Harvard scientists have transformed our understanding of olfaction from a chaotic process to a precisely engineered system. By revealing the hidden stripes of smell receptors and the chemical mechanisms that form them, this research provides a crucial foundation for future medical breakthroughs. As researchers now turn their attention to human subjects, the path toward restoring one of our most vital senses becomes clearer than ever.