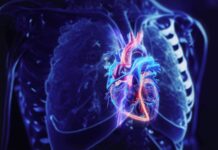
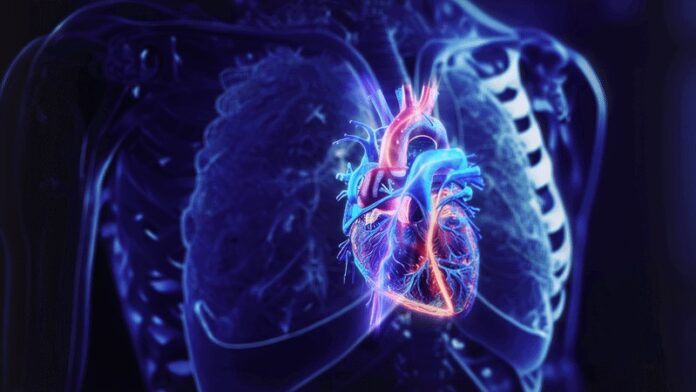
The rhythmic force of a beating heart does more than circulate blood; it actively suppresses cancer growth. A groundbreaking study reveals that the mechanical stress generated by each heartbeat triggers a specific biological response that halts the multiplication and spread of cancer cells. This discovery not only solves a long-standing medical mystery—why primary heart cancer is exceptionally rare—but also points toward a novel frontier in oncology: mechanical therapy.
Solving the Mystery of Rare Heart Cancer
Cardiac malignancies are statistically anomalies. Primary heart cancer occurs in fewer than two out of every 100,000 people annually. Even secondary heart cancer, which spreads to the heart from other organs, remains uncommon compared to metastasis in the liver, lungs, or bones.
Researchers have long suspected that the unique environment of the heart protects it from tumors, but the mechanism was unclear. The new study, published in the journal Science, identifies the culprit: mechanical load. The physical force exerted by the heart’s contraction creates an environment hostile to cancer proliferation.
The Molecular Mechanism: Nesprin-2
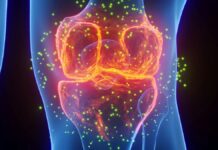
At the core of this protective effect is a protein called Nesprin-2, located in the outer membrane of the cell nucleus. Nesprin-2 acts as a biological sensor, detecting mechanical forces and translating them into genetic instructions.
The research team, led by Serena Zacchigna at the International Centre for Genetic Engineering and Biotechnology (ICGEB) in Italy, discovered that Nesprin-2 senses the force of heartbeats. When activated by this rhythmic pressure, the protein modifies epigenetic markers —chemical tags on DNA that control gene expression. Specifically, it suppresses the markers associated with tumor growth.
To confirm this, the team conducted rigorous experiments:
* In Vivo Testing: Lung cancer cells were implanted into the hearts of mice. In hearts beating normally, cancer growth was significantly restrained. In “unloaded” hearts (connected to blood flow but not pumping), cancer cells proliferated aggressively.
* In Vitro Testing: Rat heart tissue grown in lab dishes showed similar results; reducing mechanical load allowed cancer cells to spread more freely.
* Human Data: Analysis of tissue samples from patients with lung, colon, or skin cancer that had metastasized to the heart confirmed that Nesprin-2 activity correlates with suppressed tumor markers. When Nesprin-2 was disabled in beating tissue, cancer growth resumed.
“These findings are the first to show that mechanical forces beyond the tumor itself affect the growth and spread of cancerous cells,” said Zacchigna. “We saw that this signature is common to many cancer types.”
From Lab Bench to Clinical Bedside
The implications of this research extend far beyond cardiology. Since the mechanical suppression mechanism appears to be a general biological response, it could be harnessed to treat cancers in other organs.
Zacchigna and her team are developing a prototype device: elastic bands that can be strapped around skin tumors. These bands would apply rhythmic pressure to mimic the force of a beating heart, potentially triggering the same Nesprin-2-mediated suppression in metastatic skin cancer. Skin cancer was chosen as the initial target because it is one of the more common cancers to spread to the heart, making it a logical test case for this mechanical approach.
The goal is to initiate clinical trials within four years. However, significant hurdles remain:
1. Safety Validation: Researchers must ensure that squeezing tumors does not inadvertently force cancer cells into the bloodstream, increasing the risk of dissemination.
2. Patient Selection: Identifying which patients and tumor types will benefit most from mechanical stimulation requires further study.
3. Device Engineering: Creating a comfortable, effective, and safe wearable device for human use is a complex engineering challenge.
A New Paradigm in Cancer Treatment
Julie Phillippi, a cardiothoracic surgery chair at the University of Pittsburgh, described the findings as “of consequential importance.” She noted that while the immediate application may focus on non-cardiac cancers, the work also offers insights into heart tissue regeneration.
Beyond mechanical devices, the team is exploring pharmacological alternatives. If they can identify drugs that mimic the epigenetic changes caused by heartbeats, patients could receive chemical treatments that activate Nesprin-2 without the need for external pressure.
Conclusion
This research fundamentally shifts our understanding of cancer biology by highlighting the role of mechanical forces in cellular behavior. By proving that the physical act of pumping can inhibit tumor growth, scientists have opened a new avenue for therapy that could one day use simple mechanical pressure—or drugs that mimic it—to fight cancer across the body.